[463] Ming-Dong Xie, Jia-Meng Sun, Zhong-Hua Zhang, Hong-Qing Liang*, Zhi-Kang Xu*, App. Catal. B: Environ 384, 126221 (2026).
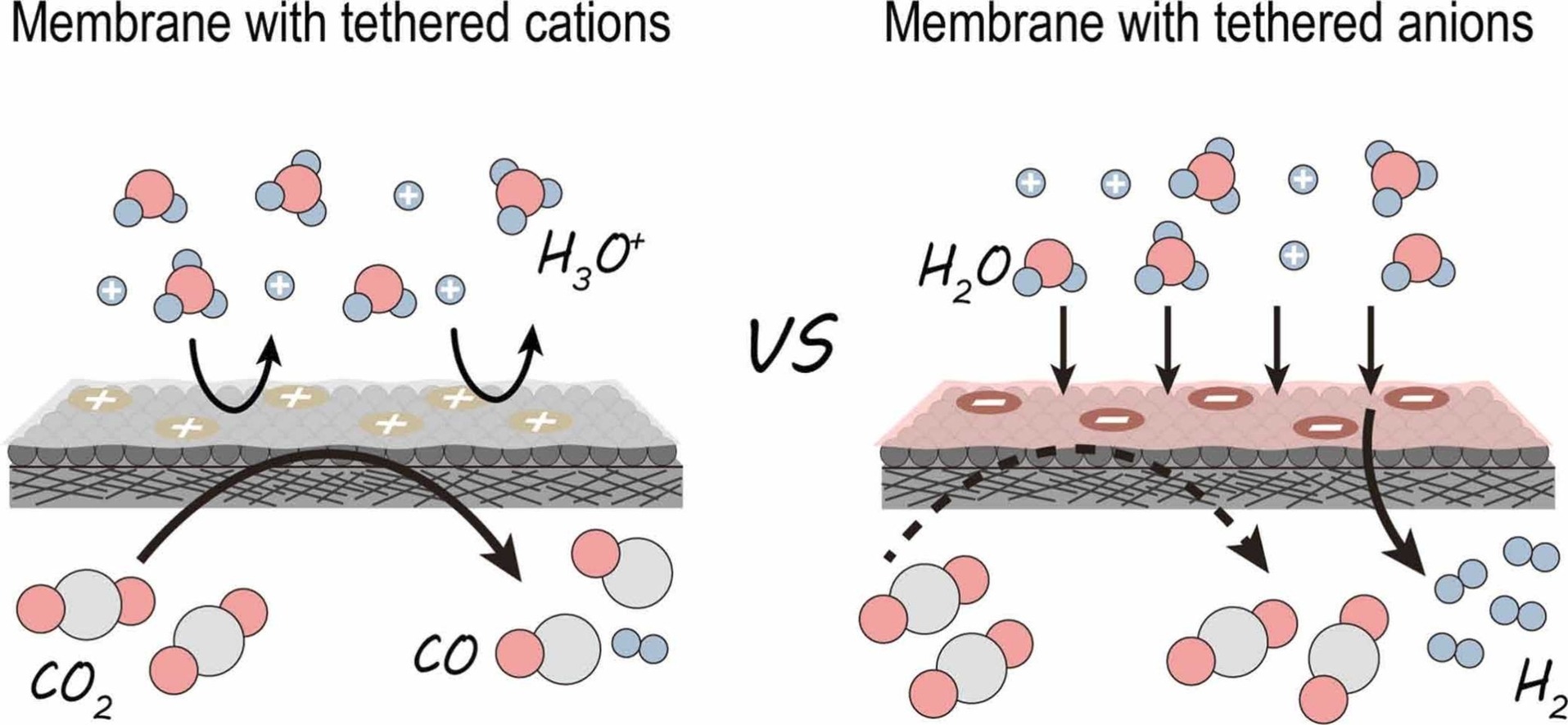
Electrochemical CO2 reduction (CO2RR) in acidic media circumvents carbonate crossover and salt precipitation inherent to alkaline and neutral systems, but suffers from low selectivity due to the dominance of hydrogen evolution. While alkali cations suppress proton activity, they inevitably lead to catastrophic (bi)carbonate deposition and electrode flooding. Here, we decouple CO2RR from alkali cations by constructing a micro-alkaline catalytic environment using a proton-blocking hydrophobic membrane with tethered cations. We designed a series of quaternized copolymers from dimethylaminoethyl methacrylate and lauryl methacrylate (qDMA-n), where the “cation–hydrophobicity balance” was precisely tuned to optimize the balance between Donnan exclusion and water management. Integrated into a zero-gap membrane electrode assembly electrolyzer, the optimized membrane enabled CO2RR on Ag catalysts with a CO faradaic efficiency of 96.9 % and a single-pass conversion efficiency of 92.4 % at 200 mA cm⁻2 in pure 0.05 M H2SO4, maintaining stable operation for over 100 h. Finite element simulations, rotating disk electrode tests and in-situ Raman spectra revealed that the balance between cationic and hydrophobic domains plays a key role in forming a stable micro-alkaline interface despite bulk acidity, leading to effective HER suppression. This work provides a practical and generalizable interface design principle for efficient CO2 electrolysis in strong acids, offering new opportunities for durable, high-efficiency CO2 electrolyzers without alkali cations.
.png)